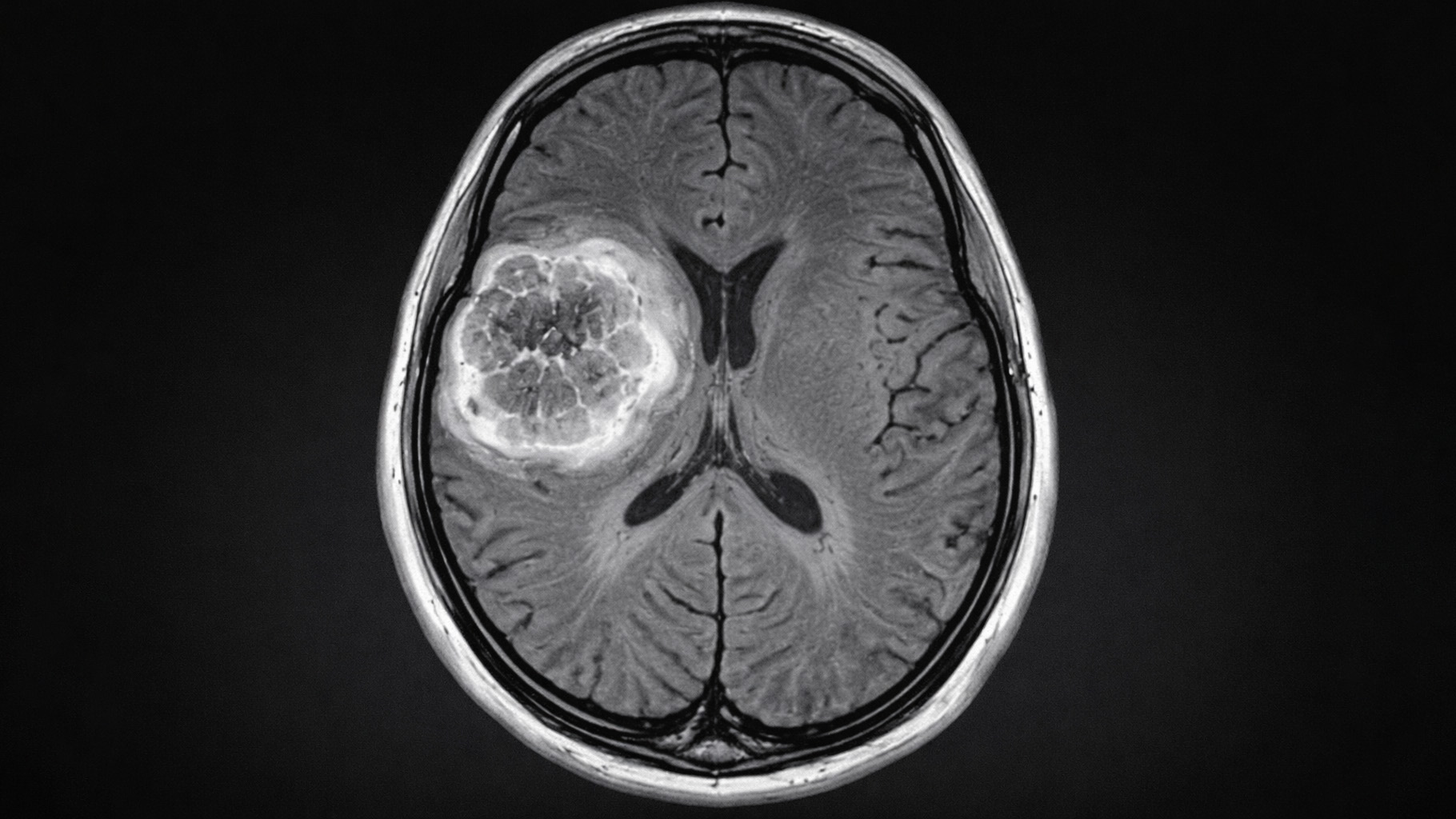
More hope in the war on cancer
“Small studies offer hope CAR-T can fight an aggressive brain cancer” reads the headline of a recent article in STAT. It includes three recent papers on the use of CAR-T to treat glioblastoma multiforme (GBM), an aggressive type of brain cancer.
There are a couple of thoughts that move to the forefront of my mind when I read titles like this on cancer treatments.
One is that the harsh realities of cancer and our lack of progress in tackling it get buried by misleadingly optimistic narratives promulgated by the news media, advocacy groups, medical centers, oncologists, cancer researchers, drugmakers, and patients, to name a few.
The other is that improving survival and quality of life while minimizing toxicity should be the aim of any rational treatment. This seemingly obvious statement tends to get lost in the shuffle of the ocean of news promising hope, progress, and breakthroughs, as well as more and more cancer drug approvals based on outcomes that don’t demonstrate a survival benefit.
I’ve adopted a way of thinking that’s a twist on a Russian proverb: Don’t trust, but verify. While I’m skeptical of titles like those above, I try not to dismiss them. Maybe it will surprise me. Some people in those same groups I just dragged through the mud are perhaps most likely to get us out of this mess. Maybe the results of the studies truly warrant optimism and suggest the researchers are on the right track. Perhaps the authors and the people they interviewed or cited address the questions that matter most to cancer patients: Will this treatment make me live longer? Will this treatment relieve my symptoms or free me from future disability? What are the adverse effects from receiving this treatment, both short- and long-term?
Before we look any further into these studies and whether they give us reason to think researchers are on the right track, we need to look at a brief history of our “war on GBM” to gain some perspective.
A brief history of GBM treatment
GBM was originally classified1They based this on the theory that the tumor stems from early glial cell precursors, known as glioblasts. The name also reflects the diverse appearance of the tumor, attributed to the presence of necrosis, hemorrhage, and cysts, hence the term “multiform.” and described in 1926 by the surgeons Percival Bailey and Harvey Cushing, documented in their 1926 monograph. At the time, they reported an average survival of 12 months in GBM patients undergoing surgery. From as early as the 1940s, clinicians have routinely used radiation to treat brain tumors, with advances in techniques and technologies over the ensuing decades. Various chemotherapies were thrown in with radiation along the way. It was not until the addition of lomustine (an alkylating agent) that survival advantages for GBM were claimed. The first cytotoxic drugs — lomustine and carmustine — were approved in 1976 and 1977, respectively. More survival advantages were to come at the turn of the century. In 1996, carmustine wafers for recurrent high-grade glioma were FDA-approved, which are biodegradable discs infused with the drug that are implanted under the skull during surgery. In 1999, temozolomide (TMZ), an oral alkylating agent that crosses the blood-brain barrier, was approved by the FDA for recurrent anaplastic astrocytoma, which is a grade 3 astrocytoma. Off-label use of TMZ for GBM (a grade 4 astrocytoma) became increasingly widespread. In 2003, carmustine wafers became FDA-approved for newly diagnosed GBM based on a trial that demonstrated the wafers plus radiation improved survival by two months compared to radiation alone. Two years later, a trial showed that TMZ in combination with radiation improved survival by two months compared to radiation alone, and the drug was granted FDA approval for newly diagnosed GBM in 2005. This established the standard of care2“The standard of care is the benchmark that determines whether professional obligations to patients have been met,” according to Donna Vanderpool in a 2021 article. “Failure to meet the standard of care is negligence, which can carry significant consequences for clinicians. … The standard of care is a legal term, not a medical term. Basically, it refers to the degree of care a prudent and reasonable person would exercise under the circumstances. State legislatures, administrative agencies, and courts define the legal degree of care required, so the exact legal standard varies by state.” for newly diagnosed GBM that is still with us today: maximum safe surgical resection followed by concurrent radiation and TMZ for six weeks, and then six monthly cycles of TMZ. Since then, another drug (bevacizumab or BVZ, 2009) and a medical device (tumor treating fields or TTFields, 2015) have been approved for treatment by the FDA.
Not only do we now have several treatments for GBM, but advocates will also point to our increased understanding of the disease, enhanced diagnostics, surgical advancements, and more nuanced treatment strategies, which have improved patient outcomes.
Given all the progress, it shouldn’t be surprising that the prognosis for someone diagnosed with GBM today is much better than it was 100 years ago.
Except, it isn’t.
Survival outcomes in GBM
“Population-level studies find median OS [overall survival] to be 8-14 months in North America,” Mostafa Fatehi and his colleagues report in their 2018 analysis of hundreds of GBM patients in addition to an evaluation of the overall literature, “which are woefully similar to numbers reported by Cushing a century ago.”
Tom Seyfried, a professor of biology, genetics, and biochemistry at Boston College and author of the 2012 book Cancer as a Metabolic Disease, recently addressed an audience at a BSI event on this topic. After listening to several presenters before him lament the replication crisis in scientific research: “Science can’t be reproduced?” he asked. “Bzzzzt! Wrong: not always: there’s nothing more reproducible than how fast people die when they’re treated with the standard of care.” Seyfried then presented a figure from Fatehi and colleagues showing survival curves from five independent surgical institutions in Canada (Figure 1).
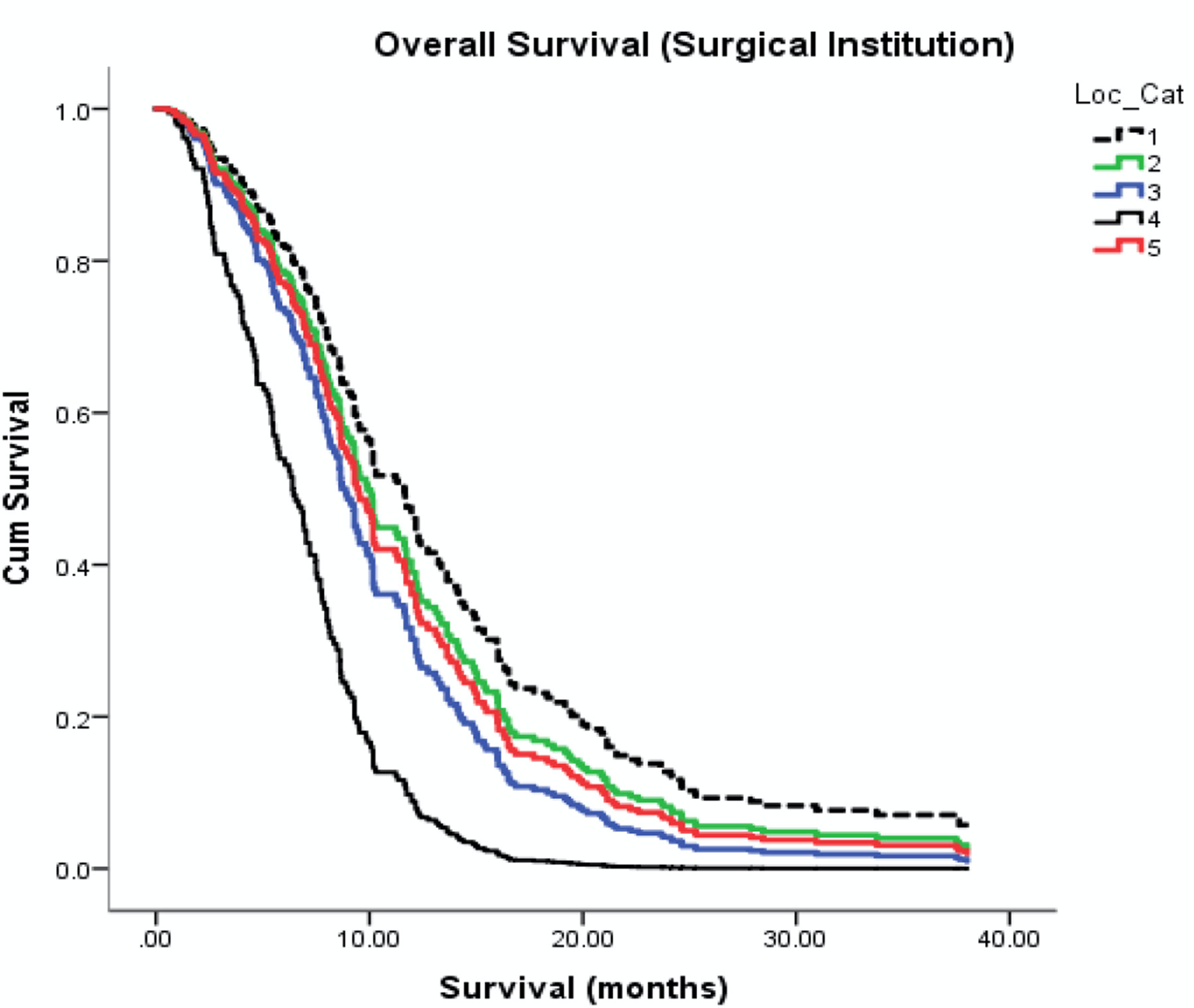
Figure 1 (Figure 3C from Fatehi et al., 2018) | Survival curves for GBM patients diagnosed from 2013 to 2015 from five Canadian surgical institutions
“I have the same survival curves from our institutions,” Seyfried says. “You’re going to get a profile that looks like this,” as Seyfried points to Figure 1. “No improvement in 100 years! … What the hell is going on with that?”
In Figure 2 below, you can see survival curves for GBM patients diagnosed from 1981 to 1991 in the US, with data from the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) Program that doesn’t look much different than the Figure 1, above.
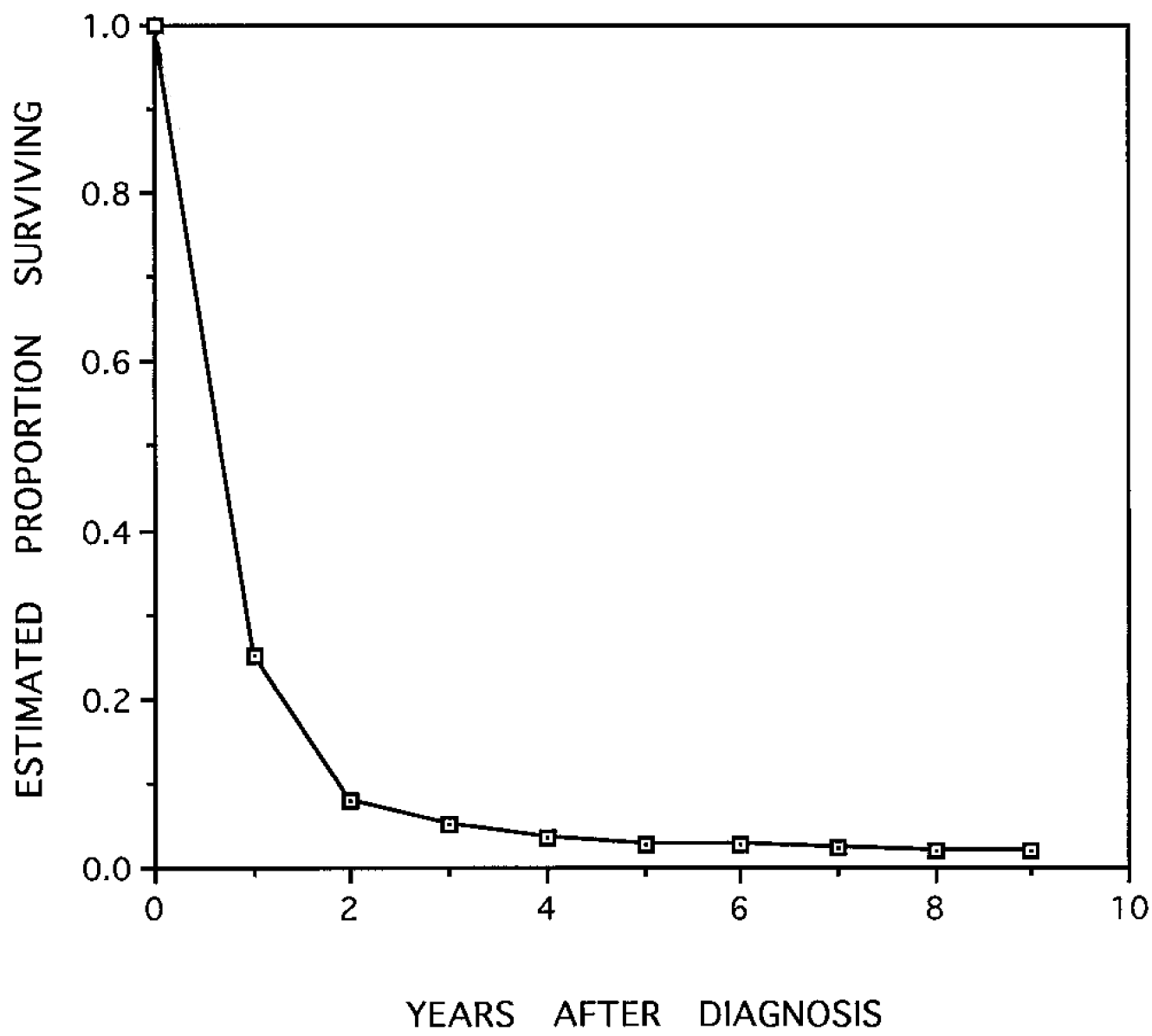
Figure 2 (from Davis et al., 1998) | Survival curves for GBM patients diagnosed from 1981 to 1991 in the U.S.
Raw data were obtained from the SEER program. Median OS ~9 mo.
In 2012, Amy Darefsky and colleagues used SEER data to demonstrate “modest, but meaningful” population-based survival improvement for GBM patients in the US following the widespread adoption of TMZ starting in 1999, as shown in Figure 3. From 1993 to 1995 and 2005 to 2007, median survival increased from 7.5 to 9.5 months.
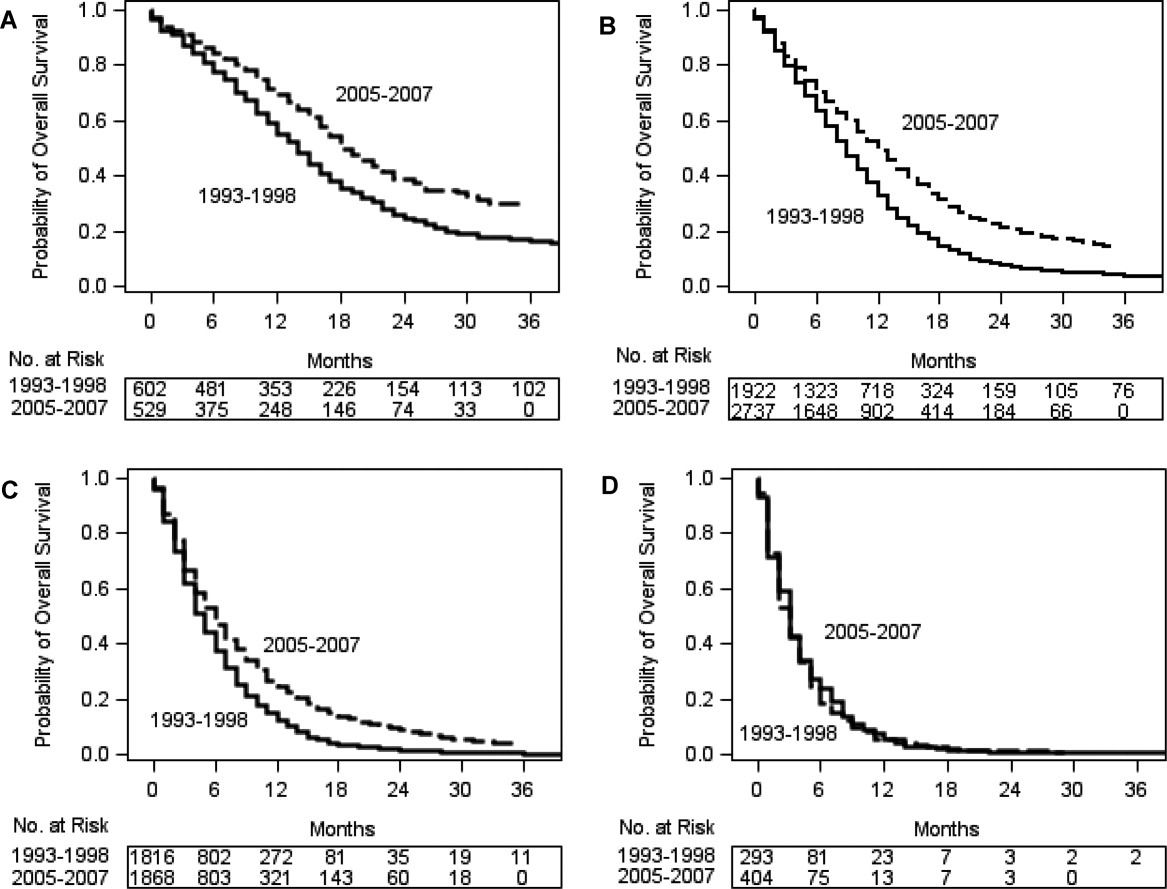
Figure 3 (from Darefsky et al., 2012) | Kaplan-Meier survival plots for glioblastoma multiforme cases according to year of diagnosis and age
Age group: (A) 20 to 44 years. (B) 45 to 64 years. (C) 65 to 79 years. (D) 80+ years. Median OS from 1993 to 1995 and 2005 to 2007, 7.5 months to 9.5 months, respectively
While we can debate whether a difference in median survival from 7.5 months to 9.5 months is modest but meaningful, it’s fair to again conclude that these numbers are woefully similar to numbers reported by Cushing a century ago. How can it be that we’ve made all the progress described above against GBM, yet patients aren’t living much longer in the 2020s than they did in the 1920s?
CAR-T studies: treatment response
So, what are the new studies offering hope and progress for immunotherapy joining the fight against GBM? The STAT article discusses three recently published studies.
INCIPIENT trial
In one interim report of a phase I trial — the Intraventricular CARv3-TEAM-E T Cells in Patients with Glioblastoma (INCIPIENT) study conducted at MGH — investigators observed “dramatic and rapid” radiographic tumor regression, occurring within days after a single infusion of CAR-T therapy engineered to target the EGFRvIII tumor-specific antigen as well as the wild-type EGFR protein, in three recurrent GBM (rGBM) participants. However, the effects were transient in two of the three participants, and their tumors progressed. In one participant, however, his tumor continued to shrink and stayed that way for more than five months after his single infusion.
PENNMED trial
In another interim report of a phase I trial, I’ll refer to it as the PENNMED study, since investigators from Penn Medicine treated six rGBM patients with CAR-T therapy targeting EGFR and IL13Rα2. Tumors appeared to shrink in all six patients. However, none met the criteria for an objective radiographic response, which was the defined secondary outcome. “Although none met criteria for an objective response … [roughly a 50% or greater decrease in the size of the tumor on imaging, sustained for at least 4 weeks],” write the investigators, “tumor shrinkage of at least 30% was observed in three of six patients, and stable disease was maintained on scans performed at least 2 months after CAR T cell therapy in three of the four patients who had at least 2 months of follow-up time.”
CoH trial
In a phase I trial, investigators evaluated CAR-T therapy, engineered to target the tumor-associated antigen IL13Rα2, a product invented at City of Hope (we’ll call this the CoH trial), in 65 patients with recurrent high-grade glioma, the majority of which had rGBM. Half (29 of 58) of the patients evaluable for disease response achieved stable disease or better. Among them, two of the patients showed partial responses, and one of the patients had a complete response. All three tumors were IDH-mutated, two were grade 3, and the other was a grade 4 astrocytoma.
So, what are we to make of the impressive responses from these three trials? Does this necessarily suggest that researchers are on the right track?
Surrogate outcomes: poor predictors of meaningful outcomes
Measures of tumor shrinkage are measures of tumor response in the cancer lingo. Measures of tumor response are surrogate endpoints, stand-ins for what we really want to measure, such as increased survival and quality of life. It seems logical that if we can shrink a patient’s tumor that patient is going to feel better and live longer as a result. “If a cancer shrinks, won’t a patient feel better?” Vinay Prasad, a hematologist-oncologist and Professor in the Department of Epidemiology and Biostatistics at UCSF, asks in his book Malignant. “While it is true that patients who have a response often feel better, it is not invariably the case.” What about survival? “[T]he link between response and living longer is tenuous. Response, then, is not assurance a patient feels better, nor is it assurance a patient lives longer. Strictly speaking, it is a surrogate.”
In 2019, Prasad and his colleagues took stock of the surrogates used in oncology, which included a quantification between response rates and survival. In the metastatic setting, the goal of treatment is to improve survival and quality of life rather than eradicating any traces of tumor in the patient because the latter has been shown to be virtually impossible. The same can be said of GBM, at least in the long term. The two most common surrogates for survival and QoL in this setting are response rate and progression-free survival. Response rate is the percentage of patients whose cancer shrinks (partial response) or disappears (complete response) after treatment. PFS is the time until either progression (typically occurs when tumors grow more than 20% from their smallest size, or there are new tumors on imaging) or death, whichever comes first.
In the metastatic setting, they examined 65 validation studies of surrogate outcomes and survival, and found:
- Six (9%) had strong correlations;
- Eight (12%) had medium correlations;
- 23 (35%) had poor correlations; and
- 28 (43%) had correlations of different strengths.
When they looked specifically at PFS and survival, regardless of cancer type, they identified 83 studies, and found:
- 16 (19%) had strong correlations;
- 27 (33%) had medium correlations; and
- 40 (48%) had poor correlations.
When they looked at response rate and survival, they identified 32 studies, and found:
- None had strong correlations;
- 3 (9%) had medium correlations; and
- 29 (91%) had poor correlations.
To put it mildly, surrogates are poor predictors of how long patients live, and response rates appear to be flat-out useless.
As for surrogates predicting how well patients live, in 2018, two groups of investigators examined this relationship. In one study, Kovic and colleagues analyzed 52 articles reporting on 38 randomized trials and did not find a significant association between PFS and health-related quality of life, concluding that their findings “raise questions about the assumption that interventions prolonging PFS also improve health-related quality of life.” In the other study, Gyawali and Hwang found that “The correlation between PFS and positive QoL was low (r = 0.34).” Roughly speaking, a correlation of this magnitude suggests that little over 10% of the changes in QoL can be predicted by PFS. Put another way, it suggests that nearly 90% of what determines QoL is unrelated to PFS.
Response and progress must be done on imaging, which may not capture the true dimensions of a tumor, especially for diffuse infiltrative tumors like GBM. “We recognize the limited reliability of pathology currently in differentiating progression from pseudoprogression,” write the authors of the update to the Response Assessment in Neuro-Oncology (RANO) in 2023, “and a RANO working group is currently addressing this issue.” Not only is it difficult for an MRI to pick up unknown lesions on diagnosis, but therapies can sometimes induce false positives and false negatives, dubbed pseudoprogression and pseudoresponse, respectively. For example, pseudoprogression is believed to be prevalent soon after completion of chemo and radiation. On the flip side, BVZ has been shown to result in radiologic response rates of up to 60% after one day of treatment. Increased enhancement on a scan can be induced by treatment-related inflammation. Therefore, pseudoprogression may represent an active inflammatory response to the tumor. That might lead one to think that an immunotherapy like CAR-T is a prime candidate for inducing pseudoprogression on an MRI. Indeed, some suggest that effective immunotherapies often seem to make a tumor grow before it shrinks it, with the former getting chalked up to pseudoprogression. However, other studies have shown that acute systemic inflammatory responses don’t interfere with early imaging results. It seems hard to argue that the INCIPIENT trial, which demonstrated “dramatic and rapid” radiographic tumor regression within days after an infusion of CAR-T, is confounded by pseudoprogression. In fact, Benham Badie, the senior author of the CoH trial, told STAT the observations of CAR-T causing GBM tumors to shrink without any pseudoprogression is a unique effect and warrants study with more advanced imaging techniques.
Summary: treatment response
The takeaway here in the context of the CAR-T and GBM studies is that, while the responses seen on MRI are encouraging, we shouldn’t assume this implies the treatments improve the quantity or quality of life in the patients who receive them. We can fool ourselves in infinite ways into detecting signals where there’s mostly noise. Perhaps advanced imaging techniques and interpretation may help reduce the level of noise and give us more confidence that what we’re seeing is truly happening. But where does that ultimately get us? Everyone should be on the same page with this one: the real signals we’re after are morbidity and mortality, which are the outcomes we should measure.
CAR-T for GBM: survival outcomes
Unlike the other two reports, the CoH trial included an analysis of one of those signals: survival. The median overall survival for all patients was eight months, and 7.7 months for the 41 rGBM patients evaluable for survival, shown in Figure 4 below.
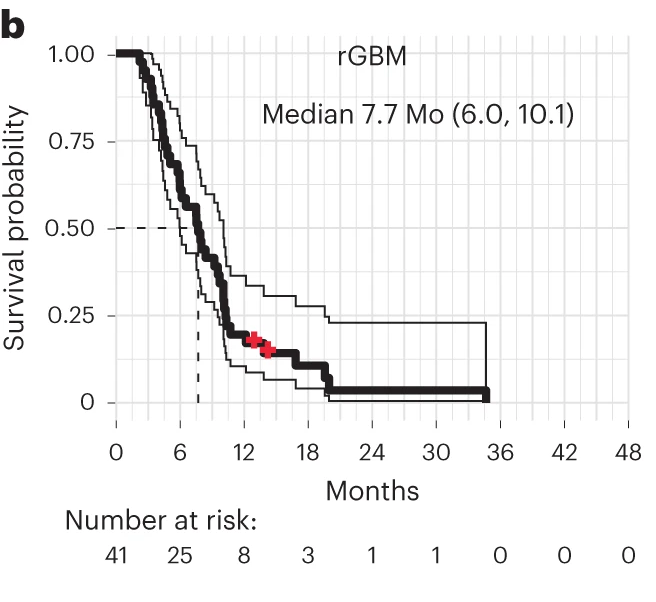
Figure 4 (from Brown et al., 2024) | Overall survival of evaluable rGBM patients from date of surgery
Thin lines denote 95% CIs; dashed line depicts median in months (Mo). Median survival with 95% CI in parentheses also indicated.
Does that look encouraging? In the light of the data on median OS for GBM of 8-14 months, 7.7 months may look particularly dismal. The results are also difficult to interpret because there was no control group in this trial. But remember, these were patients with recurrent GBM. These patients were initially diagnosed with GBM or a high-grade glioma several months before their baseline in this study, so the results are not indicative of survival for newly diagnosed GBM. (Unfortunately, this time-since-initial-diagnosis information is not provided in the paper, or I failed to find it.) Nevertheless, for a more relevant comparison, previous clinical trials have estimated a median OS in rGBM patients of about 5-13 months (McBain et al., 2021; Rostomily et al., 1994; Wong et al., 1999), so the OS results from the CoH trial appear to be nothing out of the ordinary.
Additionally, there are two important factors that likely bias the results of the CoH trial toward longer median OS compared to what might happen in the real world. The first one likely applies to all clinical trials, but the second one may be more unique to CAR-T. This is sometimes referred to as survivorship or survival bias, a phenomenon that occurs when researchers focus on individuals, groups, or cases that have made it through a selection process, and overlook those that didn’t.
Survivor bias #1: selection bias
Patients enrolled in clinical trials often have better outcomes than patients treated outside the clinical trial setting because they are intentionally selected for better prognoses. The CoH trial was no exception. Enrollment criteria included several rules of inclusion and exclusion that almost assuredly selects for relatively healthier patients. To give you a flavor, patients must exceed a threshold score (≥60%) where a higher score means the patient is better able to carry out daily activities, have a life expectancy greater than four weeks, have a limited steroid dependency, not have poorly controlled illness, and not have cardiac arrhythmias. In the real world, restrictions on who is eligible to receive the treatment may not be this rigid, were the drug to be FDA-approved. Therefore, it’s reasonable to expect decreased median OS in the real world compared to the results of clinical trials.
Survivor bias #2: The denominator problem
Two, we may not be observing the appropriate denominator of patients. The way CAR-T works is that, first, T cells are extracted from the patient’s blood. Those cells are then sent to a laboratory where they are genetically modified and cultured. Then the CAR-T cells are sent back to the clinic, where they are infused back into the patient. This takes time. In the CoH trial, the average turnaround time from cell extraction to infusion was 50 days. A couple of months is an especially long time for patients who may have a life expectancy of six months on average. Tumors progress and patients die in that window. It stands to reason that those with more aggressive tumors are more likely to progress and die, while those with less aggressive tumors are more likely to be in good enough shape to receive their treatment. The trial protocol stipulated that participants who do not receive any CAR-T infusion are considered failures of the screening process, and their enrollment ceases. Therefore, the sickest patients may have been excluded from the study. In fact, 24 patients did not receive their CAR-T therapy “due to rapid tumor progression.” Ultimately, this may have culled the sickest patients evaluable for assessing overall survival, which could have otherwise represented about 30% of this population. “The attrition before CAR T-cell infusion because of differences in disease biology between patients who remain on the study and patients who do not, a consistent theme in early-phase CAR-T trials,” argues Muhammad Abid, a professor of medicine at the Medical College of Wisconsin, “further leaves the denominator containing only those patients with biologically favourable disease, such as patients who underwent apheresis but died before cell infusion due to a greater disease burden or metabolic tumour volume.”
These factors likely inflated the number and proportion of patients achieving more favorable outcomes and are less translatable to what will happen in real-world settings. Yet, we’re still left with a median OS of less than eight months. However you slice it, it’s difficult to be encouraged by the OS reported in the trial. The senior author of the paper addressed this point head-on in the STAT article. “People lose enthusiasm looking at average survival,” Badie told STAT, “but these are very sick patients. They don’t have a lot of options. [But] these are important signals of activity.”
Summary: survival outcomes
To recap: the CoH trial seemed to show that the CAR-T treatment delayed disease progression, but it didn’t seem to advance survival. The other two reports, however small, showed remarkably rapid responses. CAR-T seemed to shrink the tumors almost instantly. But that shrinkage shown on imaging didn’t seem to last for very long, with one or two exceptions. Although, one or two exceptions out of nine patients isn’t a terrible batting average when GBM is your opponent. The more important question is whether these treatments and their tumor-shrinking capabilities on imaging translate to longer and healthier survival with minimal adverse effects. In other words, how well does tumor-shrinking on imaging predict how long or how well I live? Not very well, suggests the results from the CoH trial.
CAR-T for GBM: safety
Speaking of minimal adverse effects, what about safety? Determining the treatments’ safety was the primary objective in all three studies. These were phase I trials, which are studies intended to identify a safe dose of the treatment to take forward into subsequent trials. Determining the toxic effects and adverse events and determining the highest dose with an acceptable toxicity profile seems a logical starting point in human drug trials.
Note: All three studies used the same set of criteria for classifying adverse effects, or AEs, a grading (severity) scale from 1 through 5, so I’m including the descriptions in Table 1 below for reference.

Table 1 (from NCI, 2017) | Common Terminology Criteria for Adverse Events (CTCAE) v5.0
An Adverse Event (AE) is any unfavorable and unintended sign (including an abnormal laboratory finding), symptom, or disease temporally associated with the use of a medical treatment or procedure that may or may not be considered related to the medical treatment or procedure. Grade refers to the severity of the AE. A Semi-colon indicates ‘or’ within the description of the grade.
*Instrumental ADL refer to preparing meals, shopping for groceries or clothes, using the telephone, managing money, etc.
**Self care ADL refer to bathing, dressing and undressing, feeding self, using the toilet, taking medications, and not bedridden.
INCIPIENT trial
In the INCIPIENT trial, the primary objectives were safety and tolerability for the three participants. The investigators concluded the therapy was safe, “despite widespread expression of its target in systemic tissues.” Remember, these investigators employed a CAR-T therapy that targets the wild-type EGFR protein and a tumor-specific antigen. “The patients tolerated the infusions well,” according to a press release from MGH, “though nearly all had fevers and altered mental status soon after infusion, as was expected from an active CAR-T therapy administered into the fluid around the brain.” “We’re hopeful that if we continue to see this level of initial response, and continue to see this safety, then we’ll be able to open a larger study,” Marcela Maus, director of Mass General’s Cellular Immunotherapy Program and senior author on the paper, told Precision Medicine Online.
During the trial, investigators logged grade 3 AEs that were ‘probably related’ to treatment in two out of the three participants: severe neurotoxicity in Participant 1 and severe fatigue in Participant 3. Those weren’t the only AEs. Among the three participants over a 30-day period, investigators logged the following number of AEs at least possibly attributable to the treatment:
- 2 grade 3 AEs
- 24 grade 2 AEs
- 31 grade 1 AEs
A couple of months after Participant 1 finished his treatment in the study, he died “from disease progression,” according to the investigators, which they did not attribute to CAR-T. The cause of death was gastrointestinal perforation while he was receiving BVZ (an antiangiogenic agent) and dexamethasone (DEX, a corticosteroid). How does disease progression from brain cancer cause GI perforation? It doesn’t necessarily. “Of tumors without abdominal involvement,” write Alba Brandes and her colleagues in a 2015 overview of the most common side effects of BVZ, “GBM incurs the highest rate of gastrointestinal perforation.” In addition, “It is likely that the concomitant use of corticosteroids might contribute to this increased incidence,” the authors caution. In other words, it’s possible that the participant’s death was due to an adverse event associated with the use of treatment. Untangling deaths due to disease progression and deaths due to treatment effects is beyond the scope of this article, but it’s worth mentioning here that the latter is often nontrivial. For example, in children diagnosed with blood cancers — which make up nearly half of all childhood cancers — treatment-related mortality accounted for nearly 60% of the deaths, according to one estimate. In their words, “more children died due to their treatment than due to progression of their disease.”
PENNMED trial
In the PENNMED trial, the primary objectives were safety and determination of the maximum tolerated dose. “Taken together,” the investigators conclude, “these first-in-human data demonstrate the preliminary safety and bioactivity of CART-EGFR-IL13Rα2 cells in rGBM.”
During the trial, all six patients experienced substantial early-onset neurotoxicity, and one patient experienced a dose-limiting toxicity of grade 3 anorexia, generalized muscle weakness, and fatigue.
The investigators provided a summary of the experience of the six patients, which is worth reproducing in full.
Patient 1
Patient 1 developed grade 2 CAR neurotoxicity approximately 10 h after CAR T cell administration (day 0), characterized by increasing confusion, acute worsening of chronic aphasia [failure to understand or produce language] and nausea and vomiting. Dexamethasone and anakinra (IL-1R antagonist) were initiated with improvement in the patient’s neurologic status by day +2.
Patient 2
Patient 2, who was noted on the day −1 MRI to have rapid tumor progression, developed acute worsening of chronic left facial weakness and aphasia on day +1, consistent with grade 3 CAR neurotoxicity and prompting treatment with dexamethasone and anakinra. Improvement in neurologic symptoms back to baseline was noted by day +2.
Patient 3
In patient 3, who entered the study with worsening leptomeningeal disease and declining performance status, a reduction in the immune effector cell encephalopathy (ICE) score3“Lee et al. published a consensus grading schema for chimeric antigen receptor (CAR) T cell therapy complications, specifically cytokine release syndrome (CRS) and neurotoxicity. The immune effector cell-associated neurotoxicity syndrome (ICANS) grading schema for neurotoxicity is derived from the immune effector cell encephalopathy (ICE) score. The ICE score measures alterations in speech, orientation, handwriting, attention, and receptive aphasia.” [Herr et al., 2020] was noted on day +1. The patient was diagnosed with grade 3 CAR neurotoxicity and treated with dexamethasone and anakinra. The patient’s level of alertness and orientation waxed and waned over the subsequent 24 h, ultimately culminating in the patient becoming difficult to arouse verbally and being transferred to intensive care. Intubation was not required. Improvement in mental status was noted on day +4 with continued supportive care, and the patient returned to pre-CAR T cell neurologic baseline by day +7.
Patient 4
At dose level 2, patient 4 also experienced early and severe (grade 3) CAR neurotoxicity on day +1. The patient was initiated on dexamethasone and anakinra on day +1, and a single dose of tocilizumab (anti-IL6R) was added on day +2 in the setting of grade 2 CRS4“The CTCAE v4.03 defines CRS as ‘a disorder characterized by nausea, headache, tachycardia, hypotension, rash, and shortness of breath; it is caused by the release of cytokines from the cells.’ Although inclusive of many of the features of immune effector cell-associated CRS, this definition does not include fever, the hallmark of immune effector cell-associated CRS. CTCAE v5.0 refined the definition as ‘a disorder characterized by fever, tachypnea, headache, tachycardia, hypotension, rash, and/or hypoxia caused by the release of cytokines.’” [Lee et al., 2019] [cytokine releasing syndrome] and continued grade 3 neurotoxicity. The patient’s mental status improved markedly by day +3 and returned to pre-CAR T cell neurological baseline by day +4.
Patient 5
Patient 5 entered the study with considerable progression of multifocal tumor noted on the day −1 MRI scan. On day +1, the patient experienced grade 2 CAR neurotoxicity. The patient received only dexamethasone for supportive care, with neurotoxicity improving to grade 1 by day +2. However, on day +14, the patient presented in the setting of a dexamethasone taper with worsening fatigue, generalized muscle weakness and anorexia, each of which was grade 3 and lasted 8 d, 8 d and 14 d, respectively, meeting protocol-defined criteria for DLT. The patient’s dexamethasone dose was increased with improvement of these toxicities to grade 2 or lower by day +28.
Patient 6
Patient 6 had tumor progression involving the left midbrain and associated severe right-sided hemiparesis [muscle weakness or partial paralysis on one side of the body] on the day of CAR T cell injection. On day +1, the patient developed acute worsening of chronic expressive aphasia and further increase in right-sided weakness that progressed to complete hemiplegia [total paralysis on one side of the body]; the patient also developed aphasia on day +2, consistent with grade 2 CAR neurotoxicity, and was treated with dexamethasone and anakinra. His aphasia and ICE scores improved to baseline by day +3, but his dense hemiparesis continued. To optimize rehabilitation potential and reduce corticosteroid exposure, the patient received a single dose of bevacizumab (7.5 mg kg−1 intravenously) on day +6 and was discharged on a dexamethasone taper. There was mild improvement in right leg strength but no improvement in right arm strength by the day +28 visit.
CoH trial
In the CoH trial, the primary objectives were safety and feasibility, maximum tolerated dose, and a recommended phase 2 dose plan. “In summary,” the investigators conclude, “primary objectives of this phase I clinical study were met, establishing feasibility and safety of locoregionally delivered IL-13Rα2-CAR-T cells for treatment of [recurrent high-grade glioma] and rGBM,” the investigators conclude. And, “as there were no dose-limiting toxicities across all arms, a maximum tolerated dose was not determined.”
During the trial, grade 3 and above toxicities with possible or higher attribution to CAR-T were observed in more than a third (35%) of patients, including one grade 3 encephalopathy and one grade 3 ataxia with probable attribution to CAR-T. The most common toxicities with possible or higher attribution to CAR-T cells were headache, fatigue, and hypertension. Two patients experienced transient grade 4 (i.e., life-threatening) cerebral edema shortly after receiving their first infusion with possible attribution to CAR-T, and their symptoms improved within a few days of increasing dexamethasone (16 mg and 36 mg per day).
Summary: Safety
If you take it from the investigators and the press releases, the studies demonstrated that the CAR-T treatments were safe and well-tolerated. Given what patients go through with these treatments, those are clearly relative terms. In the protocol of the INCIPIENT trial, for example, investigators are not required to report grade 4 “expected” AEs to the IRB. Included among the expected toxicities of treatment are cytokine release syndrome (CRS) and neurological toxicity, termed ICANS (Immune Effector Cell Associated Neurotoxicity). Look at Table 2 to see what grade 4 ICANS looks like.
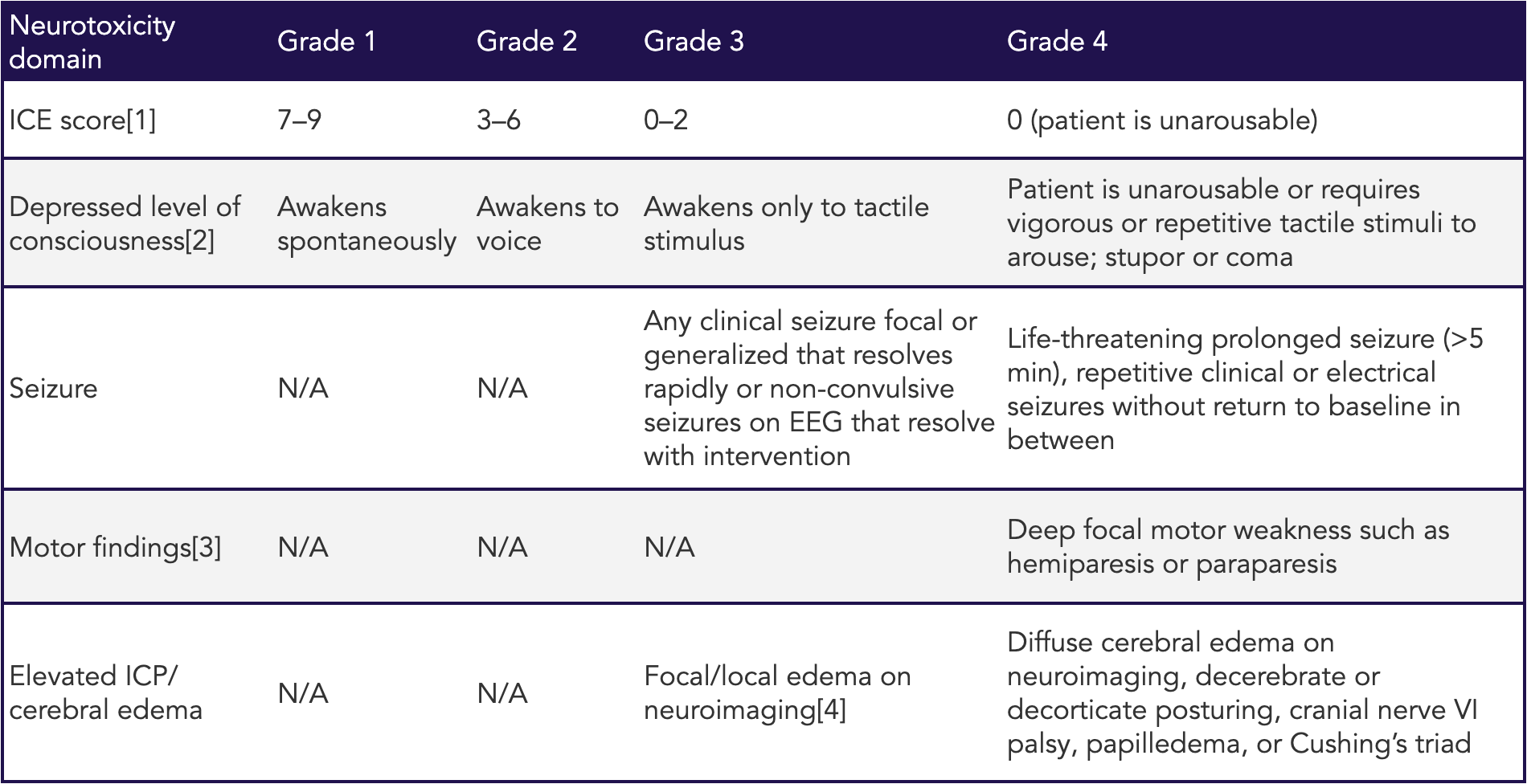
Table 2 (from Maus et al., 2020) | ASTCT ICANS consensus grading for adults
ICANS grade is determined by the most severe event not attributable to any other cause.
[1] A patient with an ICE score of 0 may be classified as grade 3 ICANS if awake with global aphasia, but a patient with an ICE score of 0 may be classified as grade 4 ICANS if unarousable.
[2] Attributable to no other cause (e.g., no sedating medication).
[3] Tremors and myoclonus associated with immune effector cell therapies may be graded according to CTCAE V.5.0, but they do not influence ICANS grading.
[4] Intracranial hemorrhage with or without associated edema is not considered a neurotoxicity feature and is excluded from ICANS grading. It may be graded according to CTCAE V.5.0.
Abbreviations: ASTCT, American Society for Transplantation and Cellular Therapy; CTCAE, Common Terminology Criteria for Adverse Events; EEG, electroencephalogram; ICANS, immune effector cell-associated neurotoxicity syndrome; ICE, Effector Cell-Associated Encephalopathy; ICP, intracranial pressure; N/A, not applicable.
“It is expected that AEs will occur frequently in this population and that these can be SAEs [serious adverse events],” the protocol from the PENNMEDICINE trial notes. “Therefore, there is no specific occurrence of SAEs that define a stopping rule, but the review of SAEs will form the basis for potential early stopping of the study.” Similarly, the CoH trial carves out a list of “anticipated” and “allowable” “expected” AEs occurring within a specified time that will not result in an expedited reporting to the FDA, nor will they result in ablation of CAR-T cells with the use of steroids.
As part of its evaluation of drugs intended to treat life-threatening and severely-debilitating illnesses, the FDA “will consider whether the benefits of the drug outweigh the known and potential risks of the drug and the need to answer remaining questions about risks and benefits of the drug, taking into consideration the severity of the disease and the absence of satisfactory alternative therapy.” Given the expected or known risks of CAR-T therapy, one would expect the benefits — increased survival and alleviation of disease symptoms — need to be substantial in order to outweigh those risks. While it’s possible this bar is cleared in future trials or future iterations of the therapy due to the clear benefits of the treatment, it can be argued that decreasing the toxicity of these treatments is just as important.
Conclusion
“Whereas most new cancer drugs afford modest benefits,” write Matthew Abola and Vinay Prasad in a 2016 article, “approved drugs or those in development may be heralded as ‘game changers’ or ‘breakthroughs’ in the lay press. … However, omission of medical context or use of inflated descriptors may lead to misunderstandings among readers.” The use of CAR-T for GBM is a prime example: “Mass General Researchers Report Major Breakthrough in Deadly Brain Cancer Treatment,” reads the title of an article from the Harvard Crimson on March 21.
It’s worth repeating: improving survival and quality of life while minimizing toxicity should be the aim of any rational treatment. Can CAR-T trials deliver on any of these for GBM? Let’s hope they can improve survival and make patients with GBM feel better, but only then will they earn the definition of breakthrough. While the investigators, fellow researchers, and media appear to be encouraged by these three small trials and believe they offer hope CAR-T can be used against GBM, their results don’t support this position from the perspective of the outcomes that matter to patients with disease. We hope that someday they do.
Part 2 – Ketogenic metabolic therapies & Advanced cancers – Coming Soon
Footnotes
Bob Kaplan is an independent research analyst. Bob previously served as director of research at Early Medical, contributing to The Drive podcast with Peter Attia and its other properties. Kaplan was a researcher at the Nutrition Science Initiative (NuSI), and an exercise physiologist at the University of Nevada, Las Vegas. His current and previous research interests include meta-research, chronic diseases, bioenergetics, exercise physiology, and nutrition.
Support the Broken Science Initiative.
Subscribe today →
recent posts
How a Low-Carb Ketogenic Diet Naturally Activates the Same Pathways
And more evidence that victory isn’t defined by survival or quality of life