Beginning in the 1950s with Ancel Keys’ lipid hypothesis, cholesterol rose to prominence as the new presumptive major risk factor for the development of cardiovascular disease. Fast-forward to today, when most everyone knows what their cholesterol usually runs, and most people probably get it re-checked every year or few.
Nowadays, of course, it’s not simply cholesterol; the report comes back with a tidy table showing the many subcategories blood lipids have been sliced and diced into—total cholesterol, VLDL, IDL, LDL, HDL, Lp(a), and triglycerides. There may be some ratios reported as well—HDL/total cholesterol, LDL/HDL, or Triglycerides/HDL that purport to refine the overall predictive value of the numbers relative to risk of a heart attack.
Of all those ratios, probably the most predictive is the triglyceride/HDL measure, which Stampfer et al. at the Harvard School of Public Health pinpointed in 1997 as the one having the strongest predictive value of who would actually go on to have a heart attack. And it’s still a worthy surrogate for who’s most at risk.
But more recently a new value has been introduced that, while still not yet standard on most lipid panels, shows up more frequently. That number is apolipoprotein B, or ApoB. And judging by the questions I received about it during a recent live Q&A on The Doctor Is In, it’s something clearly on the minds of MetFix clients, affiliate owners, managers, and staff.
What Exactly Is ApoB?
ApoB is a structural protein within the various lipoproteins mentioned above in the slice and dice array of lipid values. To better understand it, let’s begin this discussion with a quick refresher on how cholesterol gets around the body. (For those already well-versed in cholesterol synthesis and behavior, feel free to skip on down.)
Cholesterol is often thought of as a blood lipid, a blood fat, but it’s actually a waxy alcohol (its identity given away by that chemical -ol ending) that doesn’t dissolve in blood. It’s a fat-soluble molecule traveling through a water-based medium (blood), so the body loads it into carrier particles called lipoproteins. The lipoprotein is like a dump truck, and the cholesterol, its cargo. The little lipoprotein trucks trundle along in the bloodstream transporting and delivering cholesterol molecules to other lipoproteins and to tissues where they’re needed—the adrenal glands (for making cortisol) and gonads (for making testosterone, estrogen, progesterone, and DHEA) or to the liver for recycling.
Each of the major lipoprotein particles—LDL, VLDL, IDL, and other remnant particles—carries precisely one of these ApoB molecules on its surface. One ApoB per particle. Always. And that makes ApoB a fairly good tool for easily measuring the total number of all those circulating lipoprotein particles.
Your standard LDL cholesterol number, by contrast, just measures the amount of cholesterol cargo rather than the number of particles carrying it. This distinction matters, as we’ll see.
The conventional wisdom, certainly over the last couple of decades in medicine, has been that higher ApoB equals more atherogenic particles equals more cardiovascular risk. And there’s real evidence, at least on a general population-level basis, for that association. (And it should be clearly stated that at present it is really still an association, though it is frequently cast in a much more causality-proven role.)
A major meta-analysis by Sniderman and colleagues pooled data from twelve large prospective cohorts—over 233,000 people and nearly 23,000 cardiac events—and found that ApoB tracked with cardiovascular events more tightly than LDL cholesterol did. (I should divulge here that I’m not a big believer in the lipid hypothesis to start with and don’t particularly think that LDL itself is a risk factor, so that ApoB ‘tracks better than LDL’ doesn’t move my opinion much.)
The mechanistic story offered for the causal connection is that more lipoprotein particles in circulation means more opportunities for those particles to accumulate in arterial walls, which is no doubt true, but opportunity doesn’t necessarily equate with action. Still, the received medical wisdom firmly avers that the trapping of ApoB in the subintimal space is the triggering event in the inflammatory cascade that eventually becomes atherosclerotic plaque. And it may be. That’s the conventional belief, at least, and it’s not completely without merit. But it is also not the whole story.
The Lipid Hypothesis Has Real Limitations
Here it’s worth stepping back to assert something that doesn’t get said often enough in mainstream cardiology. The lipid theory of cardiovascular disease—the idea that circulating cholesterol-carrying particles are the primary driver of heart disease—remains genuinely contested.
Atherosclerosis is, at its core, an inflammatory disease. The process that turns lipid deposits into clinically dangerous plaques involves the immune system, oxidative stress, endothelial dysfunction, insulin signaling, and underlying metabolic health in ways that go far beyond particle counts. Rudolf Virchow was pointing at inflammation as a root cause of arterial disease in the mid-1800s. The lipid-centric model (thanks a lot, Ancel Keys) that came to dominate 20th-century cardiology, never fully displaced that insight; it just overshadowed it for a while.
What this means practically is that ApoB is best understood as one small signal within a much larger biological context, not as a master risk number that operates independently of everything else going on in the body. And not a number to be chased to some ‘ideal’ with an ever-expanding panoply of pharmaceuticals. How your immune system and metabolic environment interact with circulating lipoprotein particles matters enormously, and perhaps more than the particle count itself.
Not All LDL Particles Are Created Equal
This brings us to one of the most important and underappreciated findings in lipid research over the past several decades: particle differences. LDL particles vary dramatically in size, density, and biological behavior, and those differences appear to matter more than simple particle counts (ApoB numbers) or cholesterol content.
LDL particles broadly fall into two categories. Small, dense LDL particles—sometimes called Pattern B—are tightly compact, relatively cholesterol-depleted, and triglyceride-rich. (I always described them to patients as being like B-Bs, small, dense, and dangerous.) Research consistently shows that this subfraction is more prone to oxidative modification, more likely to penetrate the arterial wall, more readily taken up by macrophages, and more tightly linked to insulin resistance and the high-triglyceride/low-HDL pattern characteristic of metabolic syndrome. Studies have found small dense LDL correlates strongly with markers of systemic inflammation, such as hs-CRP (high-sensitivity C-reactive protein), and that individuals who display a Pattern B phenotype show substantially higher rates of coronary artery disease progression.
Large, buoyant LDL particles—Pattern A, or what I liked to characterize as “cotton balls”—exhibit a very different structure and behavior. Large buoyant LDL has considerably greater resistance to oxidation compared to small dense LDL, which is more susceptible due to its smaller size and higher polyunsaturated fatty acid content. LDL’s oxidative resistance is significant, because it’s oxidized and modified LDL, not native LDL, that triggers the inflammatory macrophage response central to the changes in the artery wall, where it gobbles up oxidized cholesterol and evolves into a foam cell, initiating plaque formation.
But there is also emerging and intriguing evidence about the broader biological role of LDL particles that rarely makes it into standard cardiovascular discussions.
Plasma lipoproteins including VLDL, LDL, and HDL may actually prevent bacterial, viral, and parasitic infections and are therefore a component of our innate immunity. Lipoproteins can also detoxify and neutralize lipopolysaccharide and lipoteichoic acid – bacterial endotoxins that can trigger severe systemic inflammation. In other words, LDL particles are not simply cholesterol couriers that occasionally cause trouble; they appear to play active roles in immune defense. And artificially very low levels may come with their own costs, a fact reflected in the U-shaped mortality curves seen below of large population datasets where both very high and very low ApoB are associated with higher all-cause mortality, which may help to explain the far greater mortality from Covid-19 seen in the metabolically unfit and the elderly, two groups most likely to be prescribed a statin or PCSK-9 inhibitor in search of that Holy Grail of 70 mg/dl or less on LDL-C.
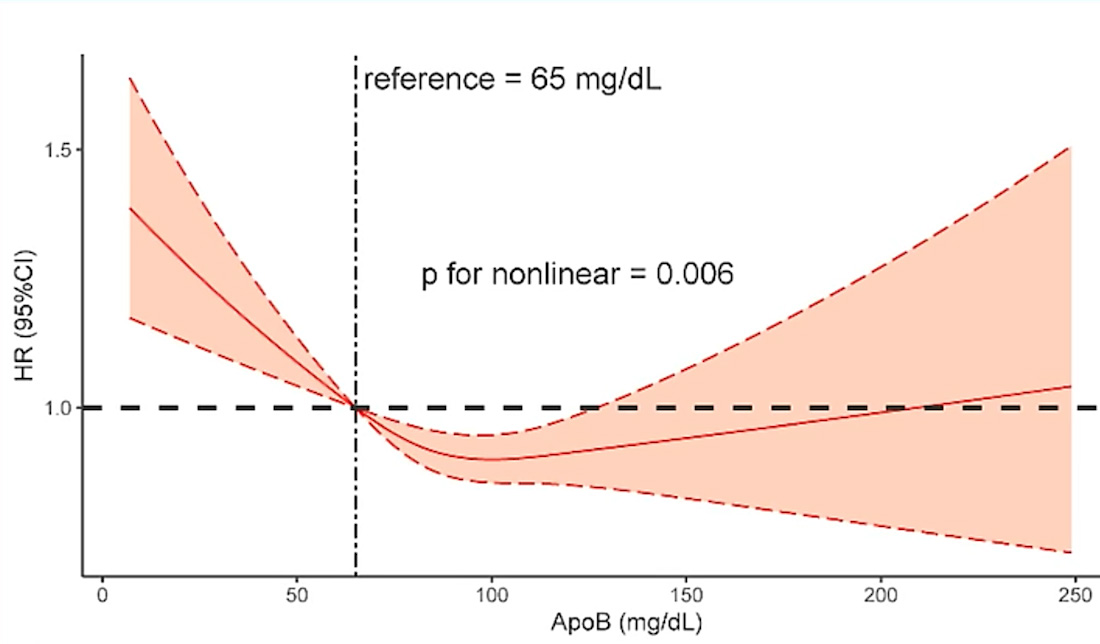
Credit: Huanqiang Li et al., Frontiers in Cardiovascular Medicine 2022 Jan 25;9:822626. Paradoxical Association Between Baseline Apolipoprotein B and Prognosis in Coronary Artery Disease: A 36,460 Chinese Cohort Study. All-cause mortality is displayed on the y-axis and ApoB level on the x-axis. (A word of caution: this is a Chinese study and even a significant percentage of Chinese scientists admit to falsifying data to get published. So grain of salt.)
The all-cause mortality data deserve a moment here. You can see from the unadjusted graph that the relationship between ApoB and all-cause mortality isn’t a straight line. It’s a U-shaped (or J-shaped) curve that has a ‘sweet spot’ of reduced mortality between about 80 and 100 mg/dl. Above and below that range the all-cause mortality risk starts to climb.
Very high ApoB does associate with higher cardiovascular death. But very low ApoB is associated with higher all-cause mortality as well, likely because very low levels reflect serious illness, malnutrition, or frailty, but also possibly because those lipoprotein particles serve genuine biological functions—including immune defense—that their absence would impair.
(According to the authors of the study, adjusting for age and nutritional status and almost every conceivable sort of disease condition including afib will flatten the upward rise on the low end to a straight descending line, implying that the lower the number the better in the exceedingly fit and healthy. But, again, it’s a Chinese study, and so maybe take the slicing and dicing that purports to show pressing ApoB lower is definitively better with a grain of salt.)
Now back to discussing the large buoyant LDL fraction that is closely tied to good metabolic health.
Diets high in saturated fats and cholesterol without question do sometimes increase the hepatic production of ApoB-containing lipoproteins, such as LDL, but in the context of a diet lower in carbohydrate that usually leads to the production of large, buoyant LDL cotton balls rather than dangerous B-Bs, and that might actually be a good thing even though it might mean more particles. (When in doubt and confronted with a rising LDL or ApoB, the determination of particle size with NMR or gel electrophoresis can often lay fears to rest on excess CVD risk.)
Conversely, high-carbohydrate diets and excessive alcohol consumption tend to promote the formation of small dense LDL particles by increasing the liver’s VLDL production. This is a finding that flips conventional dietary wisdom on its head, though the MetFix/BSI community will not likely be so surprised. The dietary pattern often blamed for high LDL-C—one higher in fat and lower in carbohydrate—actually tends to shift LDL toward the larger, less oxidation-prone variety, while the so-called heart-healthy low-fat, high-carbohydrate diet tends to generate the B-Bs that carry greater atherogenic risk.
The Fit, Metabolically Healthy Person with Elevated ApoB
All of this context becomes especially important when we consider a specific group of people, one that is growing rapidly as low-carbohydrate and ketogenic diets have become more common among health-conscious adults. And one pertinent to the MetFix community especially.
Picture someone with these numbers: triglycerides of 70–90 mg/dL, HDL above 70 or 80 mg/dL, fasting glucose in the low-normal range, fasting insulin that is low, a good Metabolic Vulnerability Score, and low markers of systemic inflammation, such as hs-CRP or GlycA.
On paper, this person looks exceptionally healthy. Their metabolic machinery is working well. They likely have large, buoyant LDL particles. And yet what if their ApoB and total LDL-C turn up modestly to moderately elevated or even a bit more? This phenomenon, sometimes called the Lean Mass Hyper-Responder phenotype, is seen frequently in lean, fit individuals on very low-carbohydrate diets. (And sometimes in those making the shift from a standard or so-called ‘heart healthy’ diet and not yet quite as lean and fit as they will be.)
Standard cardiovascular risk tools, which are largely calibrated on populations with different metabolic profiles, would flag the elevated LDL-C and ApoB as concerning. But does the standard risk calculus apply here?
The honest answer is probably not. At least not straightforwardly so, and the evidence is beginning to reflect that.
A 2025 study published in theJournal of the American College of Cardiology (JACC) examined 100 metabolically healthy individuals on long-term low-carb/ketogenic diets who had markedly elevated LDL-C, high HDL, and low triglycerides. Coronary CT angiography was used to directly measure plaque. The findings? In this specific group, traditional lipid markers including LDL-C and ApoB were not associated with baseline plaque burden or plaque progression over the follow-up period.
This is not a trivial result. It suggests that in a metabolically clean, low-inflammation biological environment, elevated circulating lipoprotein particles may not translate into the arterial wall damage that the particle-count model predicts.
And this makes biological sense when you step back and think about it. Atherosclerosis is not simply a function of how many lipoprotein particles are in your blood. It is a function of whether those particles are oxidized and modified, whether your endothelium is dysfunctional and permeable, whether your immune system is in a chronic inflammatory state, and whether your insulin signaling is impaired. A fit, metabolically healthy person with large buoyant LDL particles, excellent insulin sensitivity, low systemic inflammation, and high functional HDL is operating in a fundamentally different biological environment than a metabolically compromised person with the same ApoB number.
So How Should We Think About ApoB in This Context?
ApoB is still worth knowing. It is a more informative marker than LDL-C alone, and it tracks meaningfully with cardiovascular events in population-level data. But like all laboratory values, it needs to be interpreted in context, not as a standalone verdict.
For the person with excellent metabolic and inflammatory markers, a modest elevation in ApoB is a signal worth monitoring, not a crisis to be immediately pharmacologically managed. The relevant questions are not just “What is the ApoB number?” but a whole series of other contextual questions, such as “What is the particle composition? Are the particles large and buoyant or small and dense? What is the inflammatory environment in which they’re circulating? What does the whole metabolic picture look like? Is there evidence of actual arterial disease, such as calcium on a coronary artery calcium scan, for instance?”
A coronary artery calcium (CAC) score of zero is genuinely reassuring, particularly in someone whose other risk markers are favorable. It is one of the few tools that lets us look directly at what has actually accumulated in the coronary arteries, rather than inferring risk from blood markers. For the metabolically healthy person with elevated ApoB and LDL-C who wants to understand their actual cardiovascular status rather than their statistical risk, a CAC score is often the most useful next step. (Be aware that while a zero score is nice to have, a positive calcium score isn’t always a harbinger of serious CVD risk; there are nuances here, too. But that’s another topic for another day.)
What This Means in Practice
Cardiovascular medicine is slowly working its way toward a more contextual model of risk. The old paradigm—lower LDL-C or ApoB is always better, for everyone, regardless of metabolic context—is being refined by research that takes particle quality, inflammatory biology, metabolic health, and individual variation more seriously.
For most of the unfit and chronically ill population, elevated ApoB in the setting of high triglycerides, high LDL, low HDL, high insulin, and metabolic syndrome remains a meaningful risk signal that warrants attention. The mechanisms—small dense LDL oxidation, endothelial dysfunction, chronic low-grade inflammation—are real and well-characterized. Let me hasten to say, however, that simply artificially driving LDL-C and ApoB down with meds—meds that come with serious side effects and expense, I might add—and leaving the underlying metabolic milieu a mess isn’t likely going to be the answer to their actual problem. How much more beneficial would it be to address it with better nutrition and effective physical effort?
But for the fit, metabolically healthy individual with large buoyant LDL particles, low triglycerides, high HDL, excellent glucose and insulin control, and low inflammation, the picture is genuinely more nuanced. The early data suggest that elevated ApoB and LDL-C in this metabolic context may not carry the same atherogenic weight as identical numbers in a metabolically compromised person.
Does that mean in the context of a healthy metabolism the numbers can be safely ignored? Not quite yet, but I think we’re getting there. The studies that reassure us are still relatively short-term and small. It could be that lifelong exposure to elevated lipoprotein particle counts, whatever their size, may accumulate risk in ways that only become visible over decades, but my money isn’t on that.
Intellectual honesty requires acknowledging what we don’t yet know. But it also requires not defaulting to blanket alarm over an elevated number and chasing an arbitrary ‘ideal’ with drugs without asking what that number means in the specific person sitting in front of you. Numbers exist in bodies, and bodies have context.
The information in this article is for educational purposes and does not substitute for individualized medical advice. Please discuss your specific lipid values, risk factors, and any clinical decisions with your physician.
Physician, author, blogger, and lecturer on the art and science of low-carbohydrate nutrition, using food as a remedy for the diseases of modern civilization: obesity, diabetes, heart disease, and the myriad disorders of the insulin resistance/metabolic syndrome complex.
Support the Broken Science Initiative.
Subscribe today →
3 Comments
recent posts
How Broken Science Built the Chronic Disease Epidemic—and Why Education Is the Way Out
A primer on how modern diets disrupted human metabolic flexibility
Ketosis may support the cellular cleanup systems that decline with age


An excellent breakdown of a complex and confused topic. Thank you, Dr. Eades!
I’ll be showing this article to my GP who thankfully has referred me for this test instead of just putting me on statins!
I appreciate your pragmatic approach to this heavily nuanced topic.
One sentence, particularly resonates because it is true regardless of where the truth lies, which may be different for everyone:
“simply artificially driving LDL-C and ApoB down with meds—meds that come with serious side effects and expense, I might add—and leaving the underlying metabolic milieu a mess isn’t likely going to be the answer to their actual problem”